Best Practices for Effective Medical Device Design Reviews
.png?width=860&height=430&name=Best%20Practices%20for%20Effective%20Medical%20Device%20Design%20Reviews%20(1).png)
Design Reviews are intended to be checkpoints during medical device product development to ensure the product design is safe, effective, and progressing. Design Reviews are moments in time during design and development to ensure the device being developed is on track and in alignment with expectations.
Design Reviews are also a way to ensure Design Controls are being captured and documented throughout the project.
But I've worked on plenty of medical device projects when Design Reviews seemed like a waste of time and only took place in order to check a box for completion sake.
Still as a product development engineer back in the day, I looked forward to Design Reviews.
Why?
Design Reviews represented a time during my projects when we were one step closer to getting into the production phase and moving the product into the market. I would be sure to invite the affected functions and come prepared. I was ready to show the progress our team had made and get the approval needed to move forward.
However, my ideal Design Review scenario rarely worked out the way I had imagined. And it took me a few cycles before I could put my finger on why.
Here’s what I believe were contributing factors:
-
The company did not fully appreciate and understand Design Controls and Design Reviews.
-
I invited too many people and sometimes the wrong people.
-
I tried to cram too much into the Design Reviews.
-
I thought I was prepared, but did a poor job communicating to attendees of the Design Review agenda, purpose, and goal.
-
I expected Design Review attendees to be as knowledgeable about my medical device project as I was.
Fortunately, I had the chance to learn these lessons quickly and was able to ensure more successful and meaningful Design Reviews for the rest of my medical device product development career.
FREE DOWNLOAD: Click here to download the PDF version of this eBook.
Design controls process is not the same as the product development process
In my experience, there is a great deal of confusion about the Design Controls process and product development process.
The confusion in many companies is that Design Controls is interpreted to be synonymous with product development, but this is not the case. Design Controls "sits" within product development.

What do design reviews have to do with product development and design controls?
If a company does not understand the separation between Design Controls and product development, this confusion makes Design Reviews complicated too.
Why?
A typical product development process consists of project phases or stages. Design Controls and its deliverables do fit within specific project phases.
But remember, Design Controls are objective evidence to prove your medical device design is appropriate, safe, and effective.
When there is confusion about Design Controls and product development, Design Reviews are used as phase "gates", meaning the Design Review is used as a means to proceed to the next project phase.
It can be confusing.
Design Reviews are intended to be formal documented reviews of medical device design and development results and should be planned and conducted at appropriate stages within the Design Controls process.
Design Reviews need to be performed by individuals with an objective lens to ensure a fair analysis of the medical device design and development process.
What is a design review?
Let me share with you what FDA regulations state about Design Review from 820.30(e):
Each manufacturer shall establish and maintain procedures to ensure that formal documented reviews of the design results are planned and conducted at appropriate stages of the device's design development. The procedures shall ensure that participants at each design review include representatives of all functions concerned with the design stage being reviewed and an individual(s) who does not have direct responsibility for the design stage being reviewed, as well as any specialists needed. The results of a design review, including identification of the design, the date, and the individual(s) performing the review, shall be documented in the design history file (the DHF).
And ISO 13485:2016 in section 7.3.5 Design and Development Review states:
At suitable stages, systematic reviews of design and development shall be performed in accordance with planned and documented arrangements to:
a) evaluate the ability of the results of design and development to meet requirements;
b) to identify and propose necessary actions.
Participants in such reviews shall include representatives of functions concerned with the design and development stage being reviewed, as well as other specialist personnel.
Records of the results of the reviews and any necessary actions shall be maintained and include the identification of the design under review, the participants involved and the date of the review (see 4.2.5).
FDA and ISO are very similar on the topic of Design Reviews. The one notable difference is that FDA specifies the need for an "independent reviewer" as a Design Review attendee.
Design reviews are an essential part of the medical device design process. They are conducted during various stages of the design process to ensure that the device remains safe and complete during every stage of its development. It is the responsibility of the manufacturer and/or designer to plan and prepare for these reviews.
I realize when you read the term “stages”, the notion of phase-based and “waterfall” product development methodology may come to the surface. And if that’s your approach to medical device product development, that’s fine.
Whether your design reviews are agile in nature or more modular, both are completely acceptable methods. In fact, this is the primary intent of design reviews. A Design Review is a moment in time to compare and contrast specific aspects of the medical device. So regardless of your methodology approach, Design Reviews are still important, meaningful events.
When to do design reviews?
All Design Controls--User Needs, Design Plan, Design Inputs, Design Outputs, Design Verification, and Design Validation--need to be part of the Design Review process.
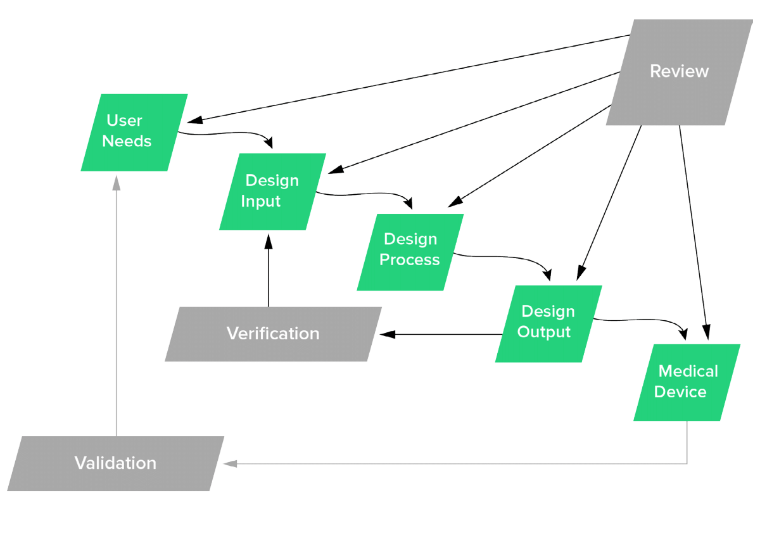
FDA Design Controls Waterfall Diagram
As far as when you should conduct Design Reviews, the fuzzy answer is that is depends on the project.
If you take the FDA Design Controls waterfall diagram shown above literally, a Design Review should happen when each step in the Design Controls process is completed. This may or may not be practical.
When you draft your Design Plan, this is the time to determine when the Reviews should take place.
Can you have too many Design Reviews in a project? To say it another way, should you take the Design Control waterfall diagram literally? That's a tough one.
If you have any questions about when to conduct Design Reviews, I would say there is no harm in having as many as possible during a project. When and how many is a function of your device and its complexity.
Design Reviews are key moments in a product development project where you can communicate status and issues of design and development. Design Reviews are moments in time to evaluate the status of design controls.
Some may even say that medical device companies are too lax with respect to Design Reviews. Here is a link to an interesting article from Cerulean LLC: "FDA, Experts Weigh in on Industry with Design Input, Design Output Activities".
The article has some good insights. I found this quote particularly interesting on the topic:
Firms have available to them the very valuable tool of design review within their quality management systems,” said Jan Welch, Deputy Director of Regulatory Affairs in CDRH’s Office of Compliance. “To the extent that design inputs are inappropriate, unachievable, poorly characterized or not documented, the design review process should identify these concerns and flag them for resolution prior to the next design steps. The same holds true for design outputs.
Design reviews should have a purpose
As the quote from the article cited above illustrates, Design Reviews should identify design and development concerns. While the quote is specific to Design Inputs and Design Outputs, realize Design Reviews serve the same purpose for ALL Design Controls. They also provide great feedback for the designer and manufacturer to consider.
One purpose of design reviews is to keep the potential device on track for development and eventual completion. By staying on track and following every step of the design process closely, you can avoid missing crucial elements of your design and catch mistakes quickly and with ease.
Another purpose of design reviews is to have an opportunity to present your potential device to potential buyers or investors. Attendees of your design reviews should include those potential buyers/investors, as well as representatives of the device’s design and development, or the design team.
As for feedback, every medical device designer needs some. Feedback, good or bad, is how you make your device better.
How many design reviews are needed?
Oddly enough, there is no good answer to this question.
It has been theorized that short, more frequent reviews are more beneficial for the device and the designer in the long run. More reviews means more opportune time for the design team to communicate concerns, ideas, and intentions.
Less frequent reviews means less communication between the designer, the design team, and the potential buyers/investors. In the process of designing a new medical device, communication is key!
At the very least, consider conducting Design Reviews for:
-
User Needs & Design Input Requirements
-
After defining Design Outputs
-
Prior to initiating Design Verification
-
After Design Verification and prior to Design Validation
-
When ready for Design Transfer
Ways to improve your design review process
I shared in the opening of this post that early on in my career I had a few not so pleasant and not so productive moments with Design Reviews.
Is there anything you can do to make sure your Design Reviews are meaningful and efficient?
Yes!
Here are 4 tips you can do use to improve efficiency in this process:
1. Strive to keep design reviews to 60 - 90 minutes
To do this, you will need to be very aware of the Design Controls you plan to cover in Design Reviews. This may mean you have to have quite a few Design Reviews during your project. It may mean that some Design Controls, such as Design Inputs, may need to be split into multiple Design Reviews.
This may seem to be quite the opposite of efficiency. But if you try to cram too much into a single Design Review, then you will defeat the purpose.
Also, really try to stick to the one hour timeframe. Any longer, and you're likely to lose a good part of your audience. Keep your design reviews timely. A good time frame for a review is 60-90 minutes. Any shorter, you won’t get through everything that needs to be discussed. Any longer, you’ll lose the attention of your audience.
2. Be prepared well in advance
Share all the documentation and information to be discussed during the Design Review with the attendees prior to the Design Review. The ideal amount of time to do this is about a week in advance, and no less than 3 business days.
Contact all intended attendees a day or two in advance of the Design Review. See what questions or comments they have and make notes. On the day of the Design Review, lead off the discussion with the comments and notes you gathered in advance.
Prepare an agenda for your Design Review. Stick to the agenda too! Don't let sidebar discussions happen. If things come up that are outside the scope of the Design Review, make a note of it and get the discussion back on track.
Be prepared. Know what you want to say and the specifics of your review. Every review should have a purpose, so it is important to stay focused and educated on each review topic. Keep an agenda of your review and stick to it.
3. Have a design review form & checklist
The whole purpose of Design Reviews is to ensure the design and development is safe, effective, and appropriate. You are reviewing specific Design Controls.
You want attendees to communicate and raise any issues or concerns. Your agenda will help guide you through the Design Review. You may also want to have a checklist prepared too, in order to make sure all required aspects are covered.
And definitely document the event on a Design Review form. At a minimum, you need to capture the topics discussed, documents reviewed, any action items identified, and list of attendees.
Be structured and organized. Keep a checklist for your review of every topic, concern, and aspect that needs to be discussed. Structure of your reviews may vary with each device you present, but the checklist should remain a part of it.
Invite your project stakeholders, including your customers. Your customers are the potential buyers/investors of your device, so it is important that they remain updated and informed on the device throughout its development. Customers are also excellent sources for feedback, good and bad.
Keep your reviews interactive and productive. No one wants to sit through a long, boring design review. Keep your audience engaged with questions and opportunities for input.
4. Close the loop for action items after the design review
During Design Reviews, there will be action items identified. It's very easy to capture these on your Design Review form.
But it's also very easy to forget about closing these action items out after the Design Review. This is a big no-no. Always close the loop for all action items.
Are design reviews bad practice?
Design reviews have gotten a pretty bad reputation on some online forums because they’re thought to be morally hazardous during the process of product development.
Why is this?
Commenters online have noticed the great increase in medical device recalls in the past decade. The design review process was implemented with the intent to reduce the number of medical device recalls, so this seems contradictory.
Despite these claims of reviews being hazardous, the opposite is more than likely true. The purpose of design reviews is to communicate and keep team members updated and informed on the development of the device.
That doesn’t sound so bad, does it?
Want to know what to avoid during your design reviews? Here are 4 not-so-good practices:
1. Don't run over your timeframe.
Long reviews are more confusing and have a negative effect on team communication. Again, keep it short and sweet!
2. Don't cram everything into a single review.
Cramming too many topics, documents, and various issues and points of discussion into one review is messy and unorganized. This is where preparing comes in to play. Prepare for your review by choosing which topics to focus on and don’t stray from your plan!
3. Don’t allow too much time between design reviews.
It is very important that your design team stays on the same page throughout the design process. By staying up to date with your team, you can avoid backtracking later in the process of development to fix little mistakes.
4. Conduct frequent design reviews.
But not so many that you lose production time. More time in review is less time spent on your device’s activity and development.
Don’t forget to document your reviews. Keep track of each of your reviews, what you spoke about, what feedback you received, and what questions were brought up in discussion. Without documentation of your review, you will likely forget important components to the furthering of your device’s development.
FREE DOWNLOAD: Click here to download the PDF version of this eBook.
Conduct digital design reviews with ease
Design Reviews are critical to the success of medical device product development. So much so that the reputable Design Controls waterfall diagram recommends that a design review should be conducted throughout every major design control stage.
At Greenlight Guru, it’s our goal to enhance your medical device product development efforts and streamline your processes through our secure, cloud-based software platform. With our medical device specific solution, companies can electronically review and approve design and development activities at multivariate stages throughout the product lifecycle. Thousands of medical device professionals across the globe are benefitting from Greenlight Guru’s purpose-built QMS Software to bring safer devices to market faster, while reducing risk. Get your free demo today ➔
Jon Speer is a medical device expert with over 20 years of industry experience. Jon knows the best medical device companies in the world use quality as an accelerator. That's why he created Greenlight Guru to help companies move beyond compliance to True Quality.
Related Posts
The Ultimate Guide To Design Controls For Medical Device Companies
Debunking 4 Commonly Held Design Control Myths
Design Reviews – Are More or Less Better?
Get your free eBook PDF
Best Practices for Effective Medical Device Design Reviews










