Greenlight Guru for Pre-Market Companies
 Calculate Your ROI
Calculate Your ROI
Get your devices to market faster with MedTech-specific solutions
Accelerate your device development with Greenlight Guru's powerful software solutions. Seamlessly integrate design controls and risk management, easily assemble regulatory submissions, and capture crucial clinical data as you prepare to launch your product.
 Calculate Your ROI
Calculate Your ROI
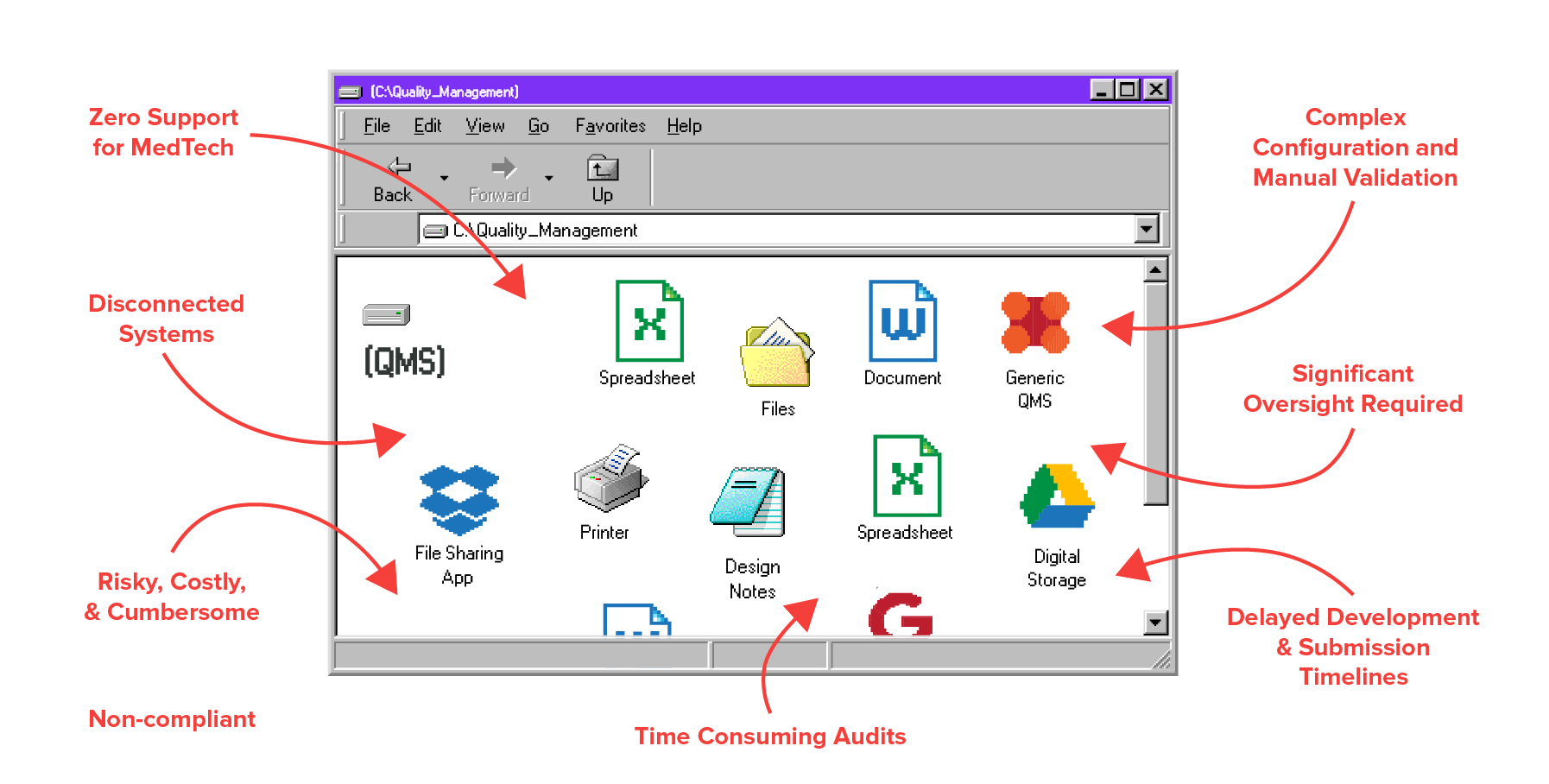
Don’t let paper slow you down.
Say goodbye to missing signatures, manual processes, and inadequate traceability. Say hello to a single, compliant system that eliminates paper and boosts efficiency across your business.
Your Old System
Greenlight Guru QMS
.png)
With Greenlight Guru, compliance and speed go hand-in-hand
Turn regulatory compliance into a competitive advantage
- Ensure compliance with software pre-validated to FDA regulations, ISO 14971:2019, and ISO 13485:2016.
- Quickly implement your QMS with more than 70 audit-tested and customizable SOP templates.
- Still have questions? With Guru Services, you have a team of industry experts on standby.
Never worry about traceability again
- Create detailed design control objects, link complex configurations, and attach documents in a few seconds.
- Manage multi-level device requirements in your traceability matrix and generate a DHF on demand.
- Stay up-to-date with Part 11 compliant e-signatures, review and approval workflows, and revision control.
Seamlessly integrate risk management and design controls
- Manage design requirements and risks side-by-side in a clean and intuitive interface.
- Get a bird’s eye view of the relationships between your requirements, risks, and verification tests.
- Build your risk acceptability matrix with personalized insights generated from real-world adverse event data.
Get the high-quality clinical data your submission needs
- Set up a study in as little as 90 seconds with a three-step study-builder.
- Collect any type of clinical data with eCRFs, ePROs, surveys, and ad hoc data collection methods.
- Ensure compliance from the start with software built to ISO 14155:2020, EU MDR, and FDA requirements.
“Greenlight Guru was the only way we could get our small team to move really quickly with a QMS.”
Monti Lejia
CEO, Delta Development Team
“As changes to the regulations occur, I don't have to worry about being out of compliance because we are using Greenlight Guru.”
Evan Luxon
Founder & CEO, Centese
“Greenlight Guru has been instrumental in helping us efficiently navigate the Quality Management System process and develop our FDA Submission.”
Ryan Nolan
Co-founder & VP of Clinical Operations, Photonicare
“Instead of taking days or weeks to prepare with a paper-based system, Greenlight Guru has allowed me to spend only a few hours preparing for an upcoming ISO Stage II audit.”
Tonia Bryant
Director of Quality & RA, Lucerno
















