Greenlight Guru for Post-Market Companies
 View Product Tour
View Product Tour
Automate your quality event workflows and stay audit-ready with MedTech-specific solutions
Ensure compliance throughout the full device lifecycle with Greenlight Guru’s software solutions. Collect and analyze post-market data, manage change effortlessly, and grow market share while driving traceability throughout your quality, design, and risk processes.
 View Product Tour
View Product Tour
Don’t let audits trip you up.
Audit findings take valuable time and energy to correct.
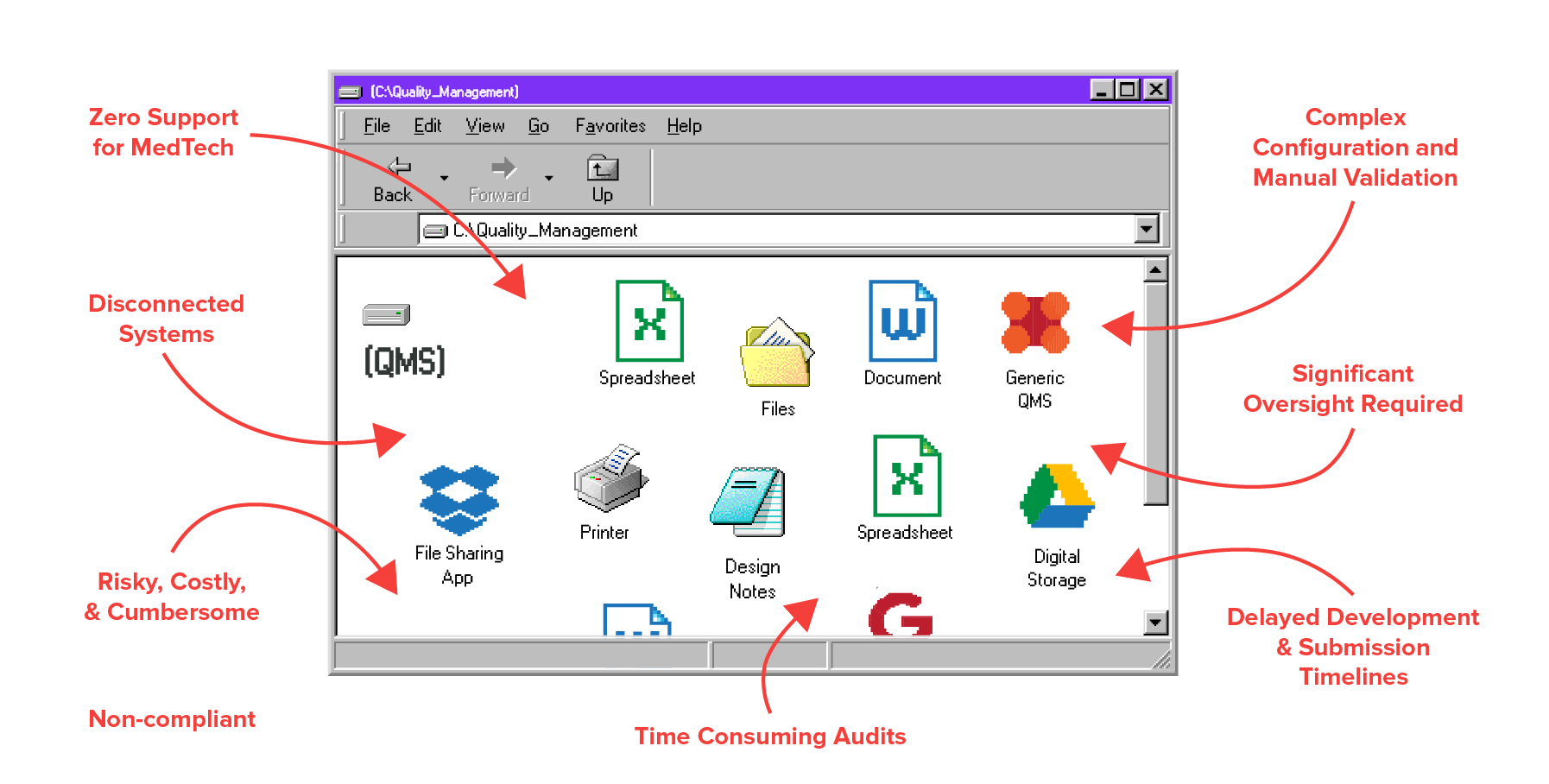
Your Old System
Greenlight Guru QMS
.png)
Get it right the first time with Greenlight Guru
Always be prepared for audits and inspections
- Work in a compliant quality system where every record is organized, centralized, and fully traceable.
- Pull any document at a moment’s notice—and know that it will always be the latest version.
- Ensure adherence to industry standards with workflows built to comply with FDA QSR, ISO 13485:2016, and EU MDR.
Proactively manage quality events with automated workflows
- Build your CAPA, Audit, Nonconformance, and Feedback processes with rapid issue resolution to enhance compliance and product improvement.
- Automate your CAPA process with custom workflows for seamless investigation, analysis, and verification activities.
- Capture, document, and analyze all types of customer feedback to quickly determine if regulatory reporting is required.
Maintain, iterate, and develop devices in a single, connected system
- Manage change effortlessly with Part 11 compliant e-signatures, flexible review and approval workflows, and revision control.
- Integrate risk management and design controls from the start, so you can quickly discover, mitigate, and prevent potential risks.
- Easily manage multi-level device requirements in your traceability matrix and generate a complete DHF on demand.
Collect high-quality clinical data throughout the device lifecycle
- Manage all your pre-market and post-market clinical activities in one connected system. Studies, surveys, ad hoc data collection—we’ve got you covered.
- Your software is already optimized for medical device studies. That means no coding, no stressful setup, and no pharma-centric features to navigate.
- Ensure compliance from the start with a system built to ISO 14155:2020, EU MDR, and FDA requirements.
- Build studies in minutes with our 3-step study builder. Copy and reuse forms or entire studies you’ve already built to save time.
“We've seen a 50% reduction in the time it takes to prepare for external audits, thanks to the streamlined workflows and essential audit templates in Greenlight Guru.”
Petra Elischer
Quality Manager, Hart Biologicals
“This was the first time in years we had no major observations in an ISO audit. We have Greenlight Guru to thank for that.”
Riley Van Hofwegen
CCO, Tenacore
“Purchasing Greenlight Guru was the best decision. Their easy-to-use eQMS, SOP Templates, and expert Guru guidance were key to us being able to successfully navigate all of our audits this year.”
Amanda Feddersen
QA Manager, Monitored Therapeutics, Inc.
“It is impossible to derail or mess up a process because Greenlight Guru has a skeleton of the workflow already built out for us. It is amazing how easy it is to link a document to a nonconformance and then back to training. It's scalable, it's repeatable, it's audit-ready.”
Alfonso Canto
QA Manager, Tenacore